The Levi laboratory in collaboration with the James laboratory is proud to present a publication in Bone Research (Nature) entitled “Neuron-to-vessel signaling is a required feature of aberrant stem cell commitment after soft tissue trauma.”
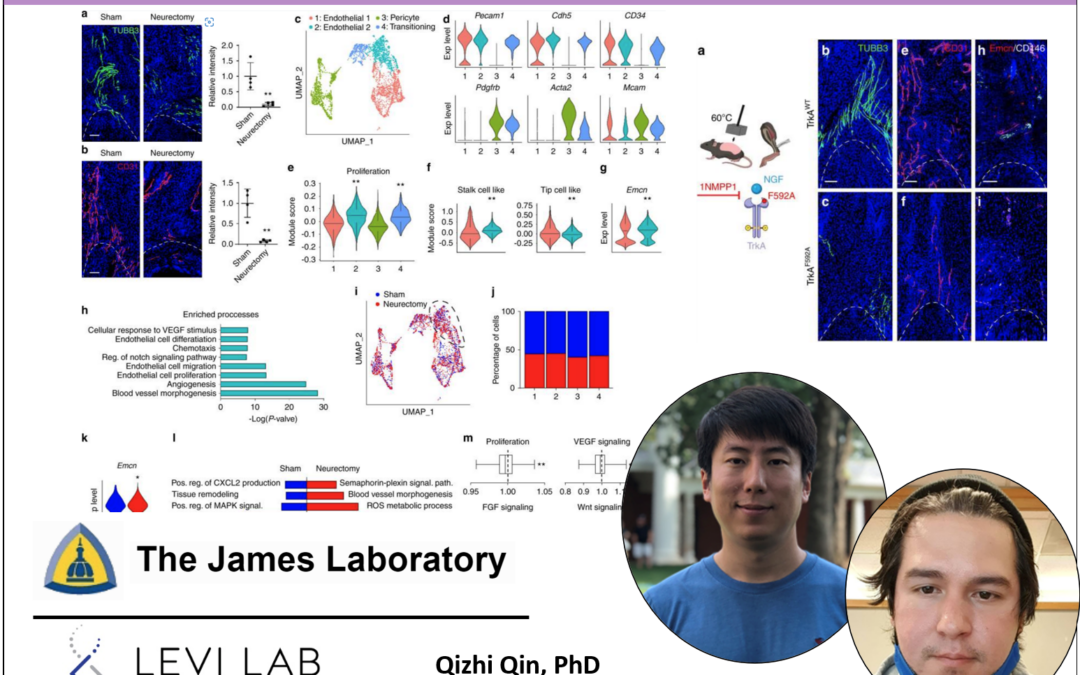
In our previous work, we previously demonstrated the independent role of sensory nerves and angiogenesis in the formation of ectopic bone formation. For the first time, we have demonstrated the interplay between both neurogenic and angiogenic programs in the aberrant formation of bone in the genesis of post-traumatic HO. In settings of neurectomy where aberrant nerve signals are profoundly inhibited, our group demonstrates changes in transcriptional programs and expression of vascular signals known to be critical regulatory axes in progression of bony deposition. These findings further propagate the paradigm of HO regulation extrinsic to endochondral ossification, whose scope was previously primarily restricted to one of the three intrinsic stages of HO formation: inflammation, mesenchymal condensation and proliferation, and chondro/osteogenesis. The research team led by Dr. Qin, Gomez-Salazar, James , and Levi of the Department of Surgery’s section of plastic surgery at Johns Hopkins University and UTSW, observed the effect of halting nerve transmission on angiogenic profiles that subsequently lead to HO formation. This work exemplifies the invaluable cross-institutional collaboration with bony experts, Aaron W. James, Seungyong Lee and Thomas Clemens, from Johns Hopkins University; Charles Hwang from Harvard University; along with the talented research teams throughout the UTSW laboratory.
PMID: 35641477. (Read here!
Abstract
The functional interdependence of nerves and blood vessels is a well-established concept during tissue morphogenesis, yet the role of neurovascular coupling in proper and aberrant tissue repair is an emerging field of interest. Here, we sought to define the regulatory relationship of peripheral nerves on vasculature in a severe extremity trauma model in mice, which results in aberrant cell fate and heterotopic ossification (HO). First, a high spatial degree of neurovascular congruency was observed to exist within extremity injury associated heterotopic ossification. Vascular and perivascular cells demonstrate characteristic responses to injury, as assessed by single cell RNA sequencing. This vascular response to injury was blunted in neurectomized mice, including a decrease in endothelial proliferation and type H vessel formation, and a downregulation of key transcriptional networks associated with angiogenesis. Independent mechanisms to chemically or genetically inhibit axonal ingrowth led to similar deficits in HO site angiogenesis, a reduction in type H vessels, and heterotopic bone formation. Finally, a combination of single cell transcriptomic approaches within the dorsal root ganglia identified key neural-derived angiogenic paracrine factors that may mediate neuron-to-vascular signaling in HO. These data provide further understanding of nerve-to-vessel crosstalk in traumatized soft tissues, which may reflect a key determinant of mesenchymal progenitor cell fate after injury.