This work showcases the extended efforts of several talented investigators including Dr. Shailesh Agarwal (Brigham & Women’s Hospital), Dr. Shawn Loder (University of Pittsburgh Medical Center), and Dr. David Cholok (Stanford University).
Furthermore, this narrative continues to exemplify the characteristic collaboration within the University of Michigan, with the project bringing together multiple experts and leaders within the fields of immunology, bone physiology, and surgery via the Knight, Mishina, Kunkel, and Ward laboratories . This work has guided the genesis of several additional targets of investigation and will continue to inform our studies on post-traumatic HO and fibrodysplasia ossificans progressive. Abstract and manuscript body forthcoming. Special thank you’s to the University of Michigan Department of Surgery and Section of Plastic Surgery, Center for Molecular Imaging, and the rest of the BWR laboratory team.
PMID: 31708911 (Read here!)
Abstract
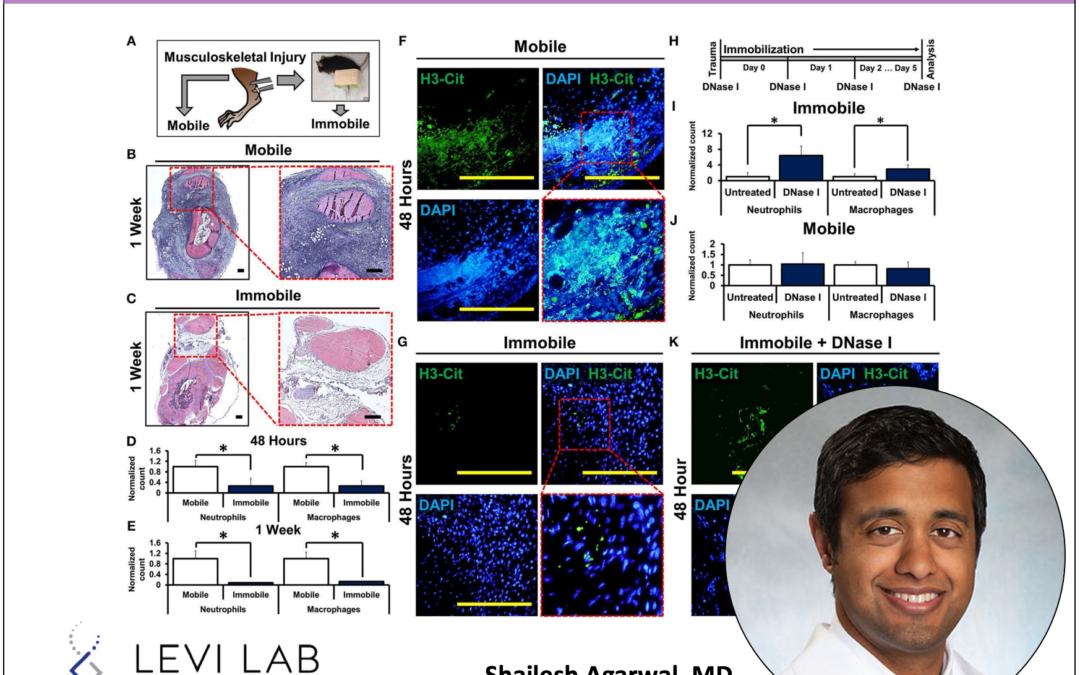
Inflammation after trauma is both critical to normal wound healing and may be highly detrimental when prolonged or unchecked with the potential to impair physiologic healing and promote de novo pathology. Mechanical strain after trauma is associated with impaired wound healing and increased inflammation. The exact mechanisms behind this are not fully elucidated. Neutrophil extracellular traps (NETs), a component of the neutrophil response to trauma, are implicated in a range of pro-inflammatory conditions. In the current study, we evaluated their role in linking movement and inflammation. We found that a link exists between the disruption and amplification of NETs which harbors the potential to regulate the wound’s response to mechanical strain, while leaving the initial inflammatory signal necessary for physiologic wound healing intact.